Water Alkalinity
Alkalinity is the ability of the water to neutralize acidity. It refers to the presence of carbonates (CO32-), bicarbonates (HCO3–) and hydroxides (OH–) and expressed in mg/L (ppm) as calcium carbonate (CaCO3). It is also known as “the buffering capacity of the water”, as it gives the water the ability to resist changes in pH upon the addition of acid.
Alkalinity = HCO3– + CO3 2- + (OH–)
The alkalinity is usually a result of weathering of carbonate rocks and dissolution of carbon dioxide. Once acid is added to water that contains alkalinity, the hydrogen ions of the acid react primarily with the carbonate species.
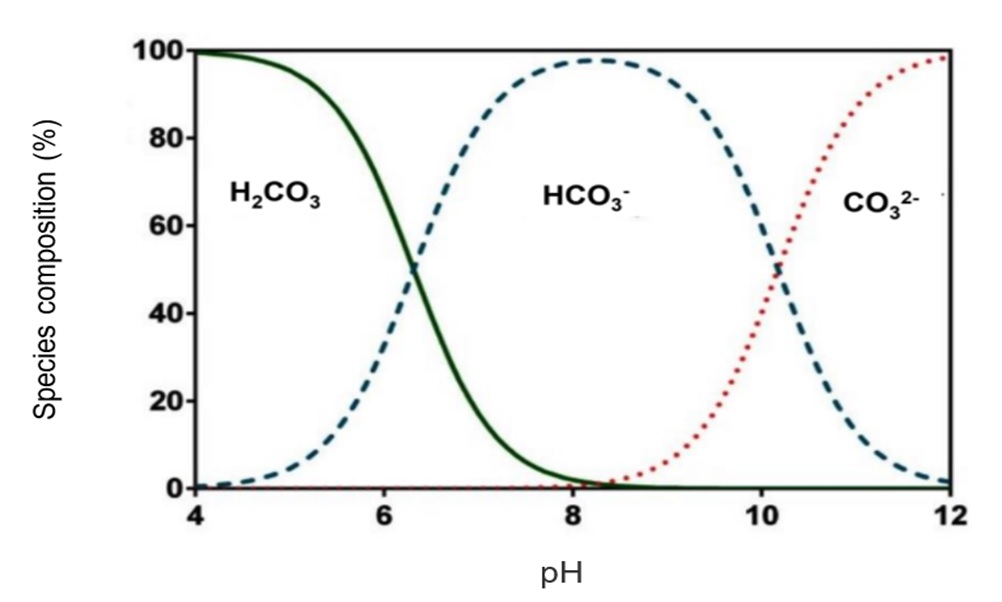
H2CO3 ↔H+ + HCO3– pH = 6.37 + log (HCO3–/H2CO3)
HCO3– ↔ H+ + CO32- pH = 10.33 + log (CO32-/HCO3–)
As can be seen in the figure above, carbonates (CO32-) exist in water at pH above 8.3 and at a pH of 4.5 it is certain that all the carbonates were converted to carbonic acid and the water does no longer have buffering capacity.
Therefore, the more alkalinity there is, the more acid is required in order to lower the water pH.
Once the alkaline compounds are consumed, any addition of acid will result in an immediate change of the pH, as the hydrogen ions, contributed by the acid, remain free in the solution.
For example, let’s compare the following two water sources:
Water source 1: pH 6.5, 120 mg/L HCO3–
Water source 2: pH 7.5, 80 mg/L HCO3-
Which water source requires more acid in order to lower the pH to 5.5?
Intuitively, one would say that water source 2 requires more acid because its pH is higher.
However, it can be seen from the titration curves below that water source 1 requires more acid in order to lower its pH to the desired level. The reason for that is the higher level of bicarbonates in water source 1 results in a higher buffering capacity, hence it is more resistant to changes in pH.